GUIDE TO INFECTION CONTROL IN THE HEALTHCARE SETTING
HOSPITAL WATER
Authors: Shaheen Mehtar, MBBS, FRCPath, FCPath, MD
Chapter Editor: Gonzalo Bearman, MD, MPH
Chapter last updated: April 2018
Print PDF
Print PDF
KEY ISSUE
- Water supply to healthcare facilities are frequently an overlooked yet essential for safe patient care and can be manageable source of infections. Numerous healthcare-associated (HAI) outbreaks have been linked to contaminated water used for patient care particularly maternal and child health, hand washing, and cleaning of medical devices for reprocessing to name but a few. Good quality potable water is still an unmet need in many low- to middle-income countries (LMIC). Infrastructure is often damaged or inadequate, leading erratic supply, contamination with fecal matter through broken pipes and wastage of water.
- Water, Sanitation, and Hygiene (WASH) programme from WHO has published guidelines for water quality, quantity, and access required in healthcare facilitiesi, and means to evaluate healthcare facilities using a comprehensive WASHFIT toolii. Safe drinking-water should meet the WHO Guidelines for drinking-water quality(2006) or national standards concerning chemical guidelines and radiological parameters and often is not available.
KNOWN FACTS
- Hospital potable water must have <1 coliform (Escherichia coli or thermotolerant) bacterium/100 mL. High levels of bacteria in hospital water, dialysate water, sinks, faucets, or shower heads has been associated with outbreaks or hand colonization.
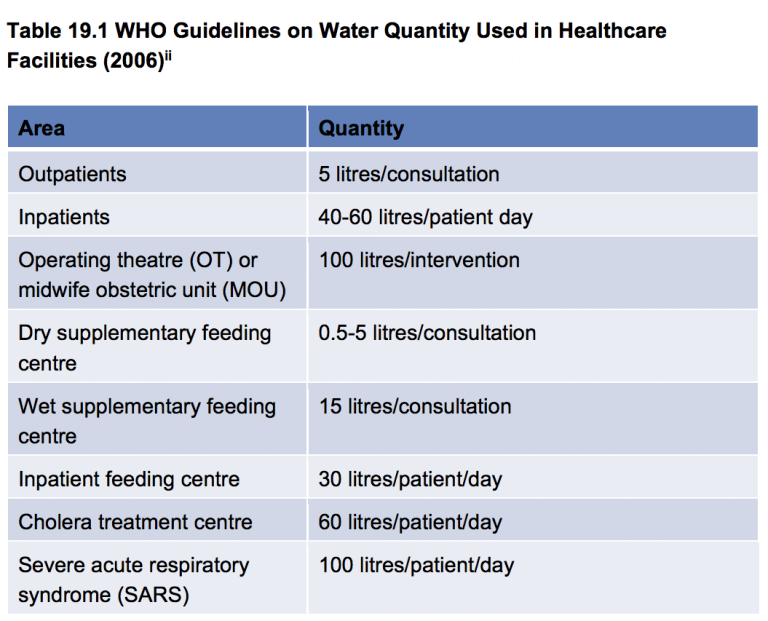
- The minimum quantities of water for healthcare facilities to carry out safe patient care must be provided (see Table 19.1)1.
- The buildup of biofilms and the corrosion of distribution lines and tank surfaces resulting from poor design or aging of distribution systems and water stagnation are the primary cause of diminished water quality.
- Colonization in more than 30% of hospital water has been associated with cases of Legionnaires’ disease. Hospital water colonization by Legionella could be long-lasting and associated periodically with outbreaks.
- Risk of illness may be influenced by several factors beside water contamination.
- In low- to middle-income countries (LMICs), high levels of water contamination correlating with low levels of chlorination have been linked to bloodstream infection outbreaks by Enterobacteriaceae, includingKlebsiella and Enterobacterspp.
- In LMICS healthcare-associated outbreaks of cholera and other waterborne diseases has been reported.
- Patient exposure to waterborne organisms occurs while showering, bathing, drinking, or with the contact of medical equipment (tube feed bags, endoscopes, respiratory equipment) rinsed with tap water.
- The implementation of a safety program for water intended for human consumption must be implemented in every healthcare facility.
Controversial Issues
- Strengthening infra structure in healthcare facilities especially in LMICs
- Maintaining high concentration of chlorine to reduce Legionella
- Routine source and point of use water testing for contamination.
- Routine point of use water filtration.
Copper may decrease water and environment colonization.
SUGGESTED PRACTICE IN ALL SETTINGS
- A high level of suspicion for cases of waterborne infections should be maintained, especially if clusters of infection occur.
- Hospital water should not be cultured routinely, however should be considered during clusters of infection particularly in LMICs.
- Handwashing sinks should be placed in the corridors or at the entrance to the patient clinical areas to reduce spread of multi drug resistant gram negative bacteriaiii. Use of alcohol-based handrubs (ABHR) should be encouraged.
- Water used for dialysis should be sampled monthly, and bacteria must be <200 bacteria/mL.
- Dialysate should be also cultured and similar levels of bacteria must be maintained.
- Nebulization devices and other semi-critical respiratory-care equipment should either be single patient use or cleaned and heat disinfected and not rinsed in tap water unless the quality of the water is known to be safe for that purpose.
- Chloride levels in hospital water should be tested periodically and the points for testing established using national or local standards.
- Use safe quality water which is either filtered or boiled for patients who are temporarily immunocompromised (e.g., bone marrow transplants)
- Where mechanical controlled environmental ventilation is provided, cooling towers should be fitted with drift reducers which direct vapour away from the hospital’s air-intake system.
- Install drift eliminators and regularly use an effective biocide to clean the water storage tanks, according to the manufacturer’s recommendations.
- In case of a single confirmed case of nosocomial Legionnaires’ disease, or two possible cases in less than 6 months, begin an epidemiological and environmental investigation. Alert hospital personnel so a high level of suspicion for the detection of new cases is maintained. This prospective surveillance should be maintained at least 2 months after the last case. If there is evidence of continuous transmission, hospital water should be sampled, and potential areas for aerosolized water should be looked. If hospital water is contaminated with Legionella, start decontamination procedures:
- Superheating: flushing outlet for at least 5 minutes with water at 65 C (149 F), (post warning signs at each outlet being flushed to prevent scald injury), or
- Hyperchlorination: >10 mg/L of free residual chlorine at point of delivery.
- Follow up cultures should be done at 2 weeks intervals for 3 months to evaluate actions taken. If no further positive cultures are found. Then cultures should be obtained monthly for another 3 months. If positive cultures are found reassess the implemented control measures, modify them accordingly, re-implement decontamination, and considerer combinations.
- In LMICs where the water supply is erratic, the infrastructure to provide water is broken or damaged, gross contamination of water is frequent. Water testing should be carried out regularly at point of use to ensure safe water supply.
Water Quality for Healthcare Facilities
- A reliable drinking-water point is accessible for staff, patients, and carers at all times.
- A reliable water point, with soap or a suitable alternative, is available at all critical points within the healthcare setting (operating theatres, wards, consulting rooms, dressing stations, etc.) and in service areas (sterilization, laboratory, kitchen, laundry, showers, toilets, waste zone, and mortuary).
- At least two handwashing basins should be provided in patient clinical areas with more than 20 beds.
- At least one shower is available for 40 users in inpatient settings (users include patients, staff, and carers staying in the healthcare setting).
- Laundry facilities, with soap or detergent, hot water and a disinfectant (such as chlorine solution), are available for inpatient settings.
Summary of Water Supply to Healthcare Facilities ii
- Water quality: Water for drinking, cooking, personal hygiene, medical activities, cleaning, and laundry is safe for the purpose intended.
- Water quantity: Sufficient water is available at all times for drinking, food preparation, personal hygiene, medical activities, cleaning, and laundry.
SUMMARY
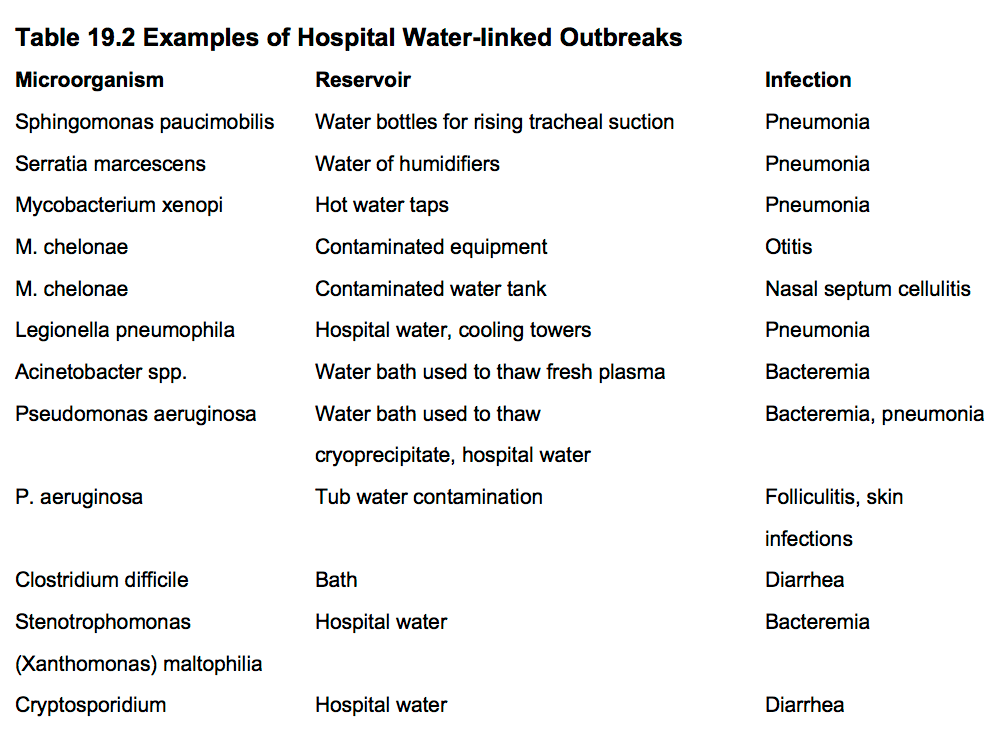
- Many bacteria can survive in water and have been linked to nosocomial infections including: Pseudomonas aeruginosa, Burkholderia cepacia, Serratia marcescens, Citrobacter freundii, Clostridium difficile, Acinetobacter baumannii, Flavobacterium meningosepticum, Aeromonas hydrophila, atypicalmycobacteria,Legionella, parasites, and virus among others. Furthermore Salmonella,Vibrio, Rotavirus, Cryptosporidium, and other enteric organisms have been reported in developing countries. In Table 19.2some examples of hospital water-linked outbreaks are shown:
- Routine cleaning, disinfection, and policies for use and changing of water from potential reservoirs should be implemented and periodically reviewed. In high-risk units the routine use of point of use filters may be a cost-effective intervention to decrease colonization and healthcare-associated infection rates.
REFERENCES
- Anaissie EJ, Penzak SR, Dignani M. The Hospital Water Supply as a Source of Nosocomial Infections. Arch Intern Med 2002; 162(13):1483–92.
- Ortolano GA, McAlister MB, Angelbeck JA, et al. Hospital Water Point-Use-Filtration: A Complementary Strategy to Reduce the Risk of Nosocomial Infection. Am J Infect Control 2005; 33(5 Suppl 1):S1–19.
- Dyck A, Exner M, Kramer A. Experimental Based Experiences with the Introduction of a Water Safety Plan for a Multi-Located University Clinic and Its Efficacy According to WHO Recommendations. BMC Public Health 2007; 7:34.
- Macías AE, Muñoz JM, Herrera LE, et al. Nosocomial Pediatric Bacteremia: The Role of Intravenous Set Contamination in Developing Countries. Infect Control Hosp Epidemiol 2004; 25(3):226–30.
- Zhou ZY, Hu BJ, Lin YE, et al. Removal of Waterborne Pathogens from Liver Transplant Unit Water Taps in Prevention of Healthcare-Associated Infections: A Proposal for a Cost-Effective, Proactive Infection Control Strategy. Clin Microbiol Infect 2013; 10:1469.
- World Health Organization. Guidelines for Drinking Water Quality (4thEdition); WHO, 2011; Geneva Switzerland; available at <http://www.who.int/water_sanitation_health/publications/2011/dwq_guidelines/en/>
- Sehulster L, Chinn RY; CDC; HICPAC. Guidelines for Environmental Infection Control in Health Care Facilities: Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC) MMWR Recomm Rep. 2003; 52(RR-10):1–42.
- Adams J, Bartram J, Chartier Y. Water Sanitation Hygiene. Essential Environmental Health Standards in Healthcare. WHO Guidelines 2008; available at http://apps.who.int/iris/bitstream/10665/43767/1/9789241547239_eng.pdf.
- World Health Organization. Water and Sanitation for Health Facility Improvement Tool (WASH FIT); ISBN 978-92-4-151169-8; WHO, 2017; Geneva Switzerland; available at
http://apps.who.int/iris/bitstream/handle/10665/254910/9789241511698-eng.pdf?sequence=1 - Hopman J, Tostmann A, Wertheim H, et al. Reduced rate of intensive care unit acquired gram-negative bacilli after removal of sinks and introduction of ‘water-free’ patient care. Antimicrob Resist Infect Control. 2017; 6:59. doi: 10.1186/s13756-017-0213-0.
